Bronchiectasis
Irreversible bronchial dilatation — from vicious cycle to surgical resection.
Irreversible dilatation of the cartilage-containing bronchi and small airways, resulting in impaired mucociliary clearance, chronic infection, and progressive parenchymal destruction.
Aetiology and pathogenesis
Cole's vicious cycle hypothesis explains the core pathophysiology: infection leads to airway insult, persistent endobronchial inflammation damages the mucociliary unit, worsening infection drives progressive bronchial dilatation. In approximately half of patients, no underlying cause is found — termed idiopathic bronchiectasis.
| Category | Conditions |
|---|---|
| Infection | |
| Bacterial | TB, Staphylococcus, Klebsiella, Pseudomonas, anaerobes, NTM |
| Viral | Adenovirus, influenza, measles, pertussis, HIV |
| Fungal | Aspergillosis (ABPA) |
| Airway obstruction | |
| Congenital | Bronchomalacia, sequestration, bronchial cyst |
| Acquired | Foreign body, tumour, lymph node compression, impacted mucus |
| Impaired secretion drainage | |
| Congenital | Cystic fibrosis, primary ciliary dyskinesia; Kartagener, Klinefelter's, yellow nail, Young's syndromes |
| Acquired | RA, Sjögren's, sarcoidosis, UC, Crohn's; toxic fume inhalation; GERD |
| Immune deficiency | |
| Primary | IgG deficiency, IgA deficiency |
| Acquired | AIDS, malnutrition |
Clinical features
Chronic productive cough with copious sputum is the hallmark. Paroxysms occur on waking due to pent-up nocturnal secretions. Sputum is thick, tenacious, and foul-smelling. Approximately half of patients experience haemoptysis, ranging from streaking to life-threatening massive haemoptysis. Dry (sicca) bronchiectasis — cough and haemoptysis without expectoration — occurs in a subset.
Always document inciting events — childhood pneumonias, TB exposure, foreign body inhalation, allergic disease, and systemic symptoms of autoimmune disorders. Extensive bronchiectasis can exist with minimal physical signs. On auscultation: early and mid-inspiratory coarse leathery crackles that reduce after coughing and postural drainage.
Imaging
HRCT — investigation of choice
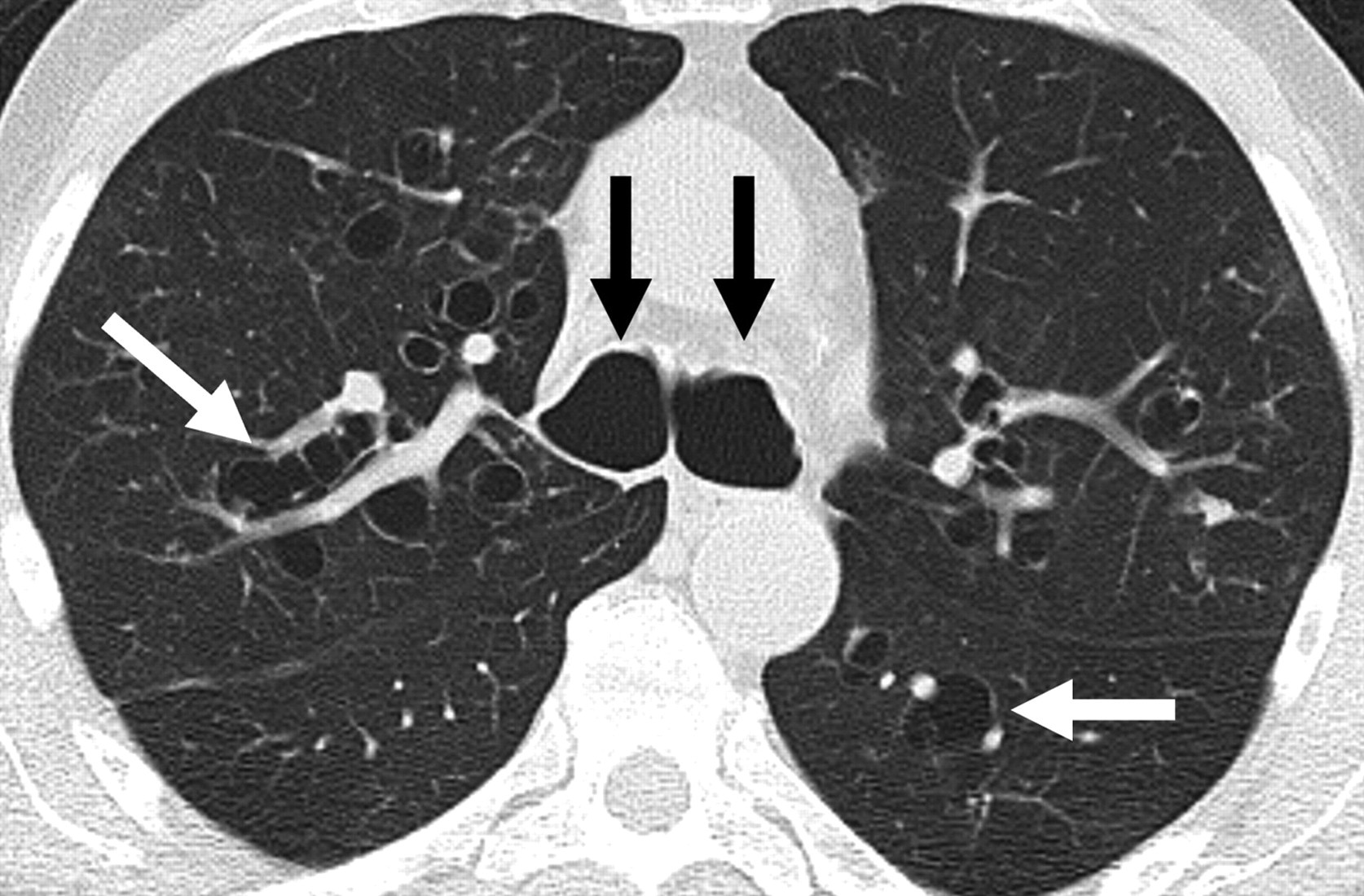
| Sign | Finding | Significance |
|---|---|---|
| Tram-track sign | Parallel thickened bronchial walls, uniform calibre, no tapering | Cylindrical bronchiectasis |
| Signet ring sign | Broncho-arterial ratio >1.5 — dilated bronchus beside normal artery | Diagnostic of bronchiectasis |
| Cluster of grapes | Dilated cystic bronchi to pleural surface with air-fluid levels | Severe cystic/saccular disease |
| Other | Mucoid impaction, air-trapping, mosaic perfusion | Functional obstruction |
Reid classification: Three morphological types in increasing severity — cylindrical (tubular), varicose, and cystic/saccular.
Surgical indications
Surgical indications have remained broadly stable since the 1950s. Localised bronchiectasis should be offered resection in intermediate and late disease. Diffuse bilateral disease carries poor prognosis with either modality.
- Frequent infective exacerbations requiring repeated antibiotic therapy
- Failure of adequate medical management
- Complications — haemoptysis, lung abscess, empyema
- Prevention of contamination spread from severely diseased to relatively normal segments
- Localised NTM disease resistant to prolonged medical therapy
Resection must be complete — leaving residual bronchiectatic segments is associated with persistent symptoms and poor outcomes. Reduce septic load pre-operatively with at least two weeks of targeted antibiotics, physiotherapy, and postural drainage before any elective resection.
Operative approaches
Standard approach. Excellent exposure. Staged bilateral resections are separated by 6 weeks.
For simultaneous bilateral resection — particularly middle lobe and lingular segments. May need clam-shell extension for LLL.
Suitable for localised disease with minimal scarring, no calcified hilar nodes. Preferred for selected single-stage bilateral resections.
Dense vascular adhesions and hypertrophied bronchial arteries demand meticulous dissection and haemostasis. Calcified nodes encasing the PA may require proximal arterial control or intrapericardial dissection. Clamp and divide the bronchus early to minimise soiling. Preserve peribronchial tissue to reduce BPF risk. Cover all stumps with vascularised tissue.
Postoperative complications
Overall morbidity approximately 20%. No significant difference between bilateral diffuse and localised disease.
For antibiotic therapy, microbiological surveillance, and non-surgical management protocols, refer to the BTS Guideline on non-CF bronchiectasis (current version).
All clinical content should be verified against current guidelines before clinical application. This resource is intended for revision and educational purposes only.
Standard textbooks
- Shields TW, LoCicero J, Reed CE, Feins RH. General Thoracic Surgery. 7th ed. Lippincott Williams & Wilkins.
- Sellke FW, del Nido PJ, Swanson SJ. Sabiston & Spencer Surgery of the Chest. 9th ed. Elsevier.
- Pearson FG, et al. Thoracic Surgery. 3rd ed. Churchill Livingstone.